Abstract
Introduction.
Older patients aged over 65 with acute myeloid leukemia (AML) are often ineligible for hematopoietic stem cell transplantation (HSCT) and generally have a poor prognosis. The personalized strategy for appropriate treatment for these patients has not yet been completely established. While C-reactive-protein (CRP) to albumin ratio (CAR) based on serum level of CRP and albumin (Alb) predicts prognosis of patients with hematological malignancies treated with HSCT (Miyashita E, BBMT. 2019), the prognostic significance of this inflammatory and nutritional assessment in transplant-ineligible elderly patients with AML remains to be clarified.
Methods.
Hokkaido Leukemia Net (HLN) is prospective cohort study collecting AML samples from hospitals of North Japan Hematology Study Group (NJHSG). In this study, we focused on older (≥65 years) patients with newly diagnosed AML treated without HSCT and investigated cytogenetic and molecular abnormality of leukemic cells including FLT3-ITD, NPM1, CEBPA, and KIT. We stratified the patients into favorable, intermediate, and adverse risk group based on NCCN 2017. Based on the result of blood sample test, we assessed CAR [CRP (mg/dL) / Alb (g/dL)] at diagnosis for each patient. Since the median CAR at diagnosis was 0.52 in this cohort, we defined CAR < 0.52 and ≥ 0.52 as CAR low and CAR high, respectively. We evaluated the impacts of NCCN 2017 and CAR on overall survival (OS) in these patients. The study procedures were in accordance with the Helsinki Declaration and institutional ethical guidelines, conducted under the auspices of the institutional ethics committee, and approved by the institutional review boards.
Results.
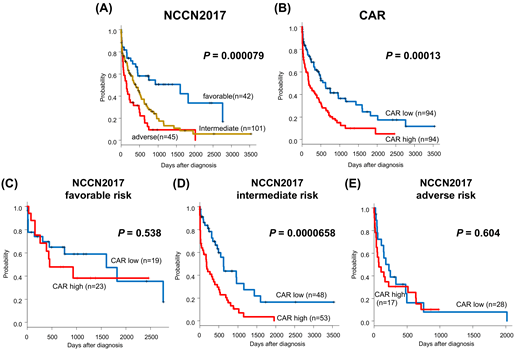
In total, 197 patients with newly diagnosed AML patients aged 65 or older enrolled in HLN between April 2010 and March 2019. Nine patients undergone HSCT were excluded, and 188 patients were reviewed (Age 65-93, median 72; male 119, female 69). In this cohort, classification based on NCCN 2017 successfully divided the prognosis of the patients for 2-year and 5-year OS [2-year OS; favorable group, 58.1%; intermediate group, 29.4%; adverse group, 12.6%, 5-year OS; favorable group, 33.7%, intermediate group, 8.74%; adverse group, 9.44%, P=0.000079, Figure A]. OS in patients with CAR 0.52 or more was significantly lower than the low score group [2-year OS; CAR low group, 43.4%; CAR high group, 21.5%; 5-year OS; CAR low group, 21.0%; CAR high group, 9.96%, P=0.00013, Figure B]. In a univariate analysis, higher age, adverse group in NCCN 2017, high CAR, and not reached complete remission (CR) after first induction chemotherapy were associated with poor 2-year OS. A multivariate analysis demonstrated that adverse group in NCCN 2017 and high CAR were independently associated with poor 2-year OS (adverse group in NCCN 2017; HR, 1.671; 95% CI, 1.157 to 2.421, P=0.007, high CAR; HR, 1.572; 95% CI, 1.093 to 2.276, P=0.01618; log-rank). Combined with NCCN 2017, while there were no significant prognostic impacts of CAR at diagnosis in patients with favorable or adverse risk groups (Figure C, E), we found that in intermediate risk group the patients with high CAR had poorer prognosis than patients with low CAR [2-year OS; CAR low group, 43.7%; CAR high group, 15.9%, P=0.0000658; Figure D]. Altogether, we demonstrated that high CAR predicts poor prognosis in the transplant-ineligible elderly patients with newly diagnosed AML and improves risk stratification of the patients with NCCN 2017 intermediate risk group.
Conclusion.
CAR is a simple and easily evaluable parameter for predicting outcomes of transplant-ineligible elderly patients with newly diagnosed AML, independently from NCCN disease risk classification. Moreover, CAR can further stratify the prognosis of patients with NCCN 2017 intermediate risk group and improve the predictive risk stratification for elderly AML patients.
Kondo: SANWA KAGAKU KENKYUSHO CO.,LTD.: Consultancy; Astellas Pharma Inc.: Consultancy, Honoraria; Otsuka Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Novartis Pharma KK: Honoraria; Bristol-Myers Squibb Company: Honoraria; Sumitomo Dainippon Pharma Co., Ltd.: Honoraria. Teshima: Merck Sharp & Dohme: Membership on an entity's Board of Directors or advisory committees; Nippon Shinyaku Co., Ltd.: Research Funding; Pfizer Inc.: Honoraria; Fuji pharma CO.,Ltd: Research Funding; Kyowa Kirin Co.,Ltd.: Honoraria, Research Funding; TEIJIN PHARMA Limited: Research Funding; Bristol Myers Squibb: Honoraria; Janssen Pharmaceutical K.K.: Other; CHUGAI PHARMACEUTICAL CO., LTD.: Research Funding; Astellas Pharma Inc.: Research Funding; Takeda Pharmaceutical Company: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis International AG: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; Gentium/Jazz Pharmaceuticals: Consultancy; Sanofi S.A.: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal